affordability versus Access: How can we find a fair system for everyone?
1. The trade-off between regulation and access
2. What is the Willingness‑to‑Pay for pharmaceuticals?
3. Does EU membership improve access to new medicines?
1. Who regulates the pricing of new drugs?

Photo: HCHE
ANMOG
In 2011, The German Pharmaceutical Market Restructuring Act ("AMNOG", in German: Arzneimittelmarktneuordnungsgesetz) introduced a process to determine the price of new pharmaceuticals.

Photo: HCHE
AMNOG has slightly increased launch time
The introduction of ANMOG has increased the time it takes for new drugs to be launched in Germany.
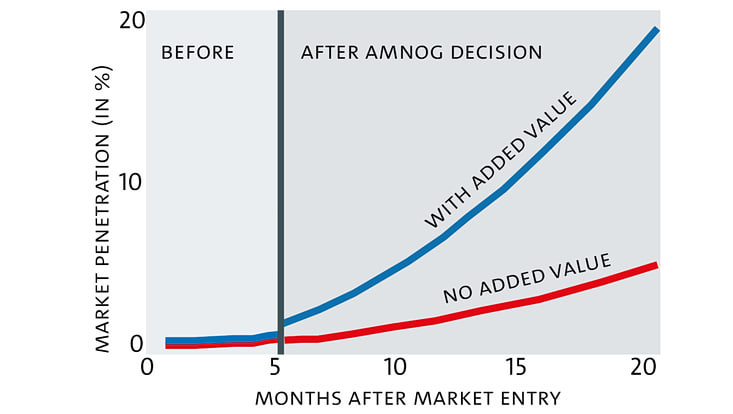
Photo: HCHE
Market entry is slower, but what are the benefits?
"A positive assessment is like a quality rating. Physicians are more likely to prescribe pharmaceuticals with added value. So, it speeds up diffusion and could compensate for the effects of a delayed launch."
Prof. Tom Stargardt
2. What is the willingness‑to‑pay for pharmaceuticals in Germany?

Photo: HCHE
Pricing is controversial
Germany has the largest market for pharmaceuticals in Europe and the third largest worldwide. It is characterized by high availability of drugs and rapid reimbursement. By international standards, spending on pharmaceuticals is high and has increased significantly in recent years, highlighting the need to take a closer look at prices.

Photo: HCHE
Willingness-to-pay for newly-authorized pharmaceuticals
We examined willingness-to-pay (WTP) with regard to newly-authorized pharmaceuticals for three major diseases: diabetes, cardiovascular disease and psoriasis.

Photo: HCHE
Value may differ by treatment
The value of treatments varies depending on how health benefits from rare or severe diseases are weighted and on the higher willingness-to-pay for innovations. In addition, small patient populations and complex manufacturing processes – such as those involved in biologics – justify a higher price.
3. Can the EU act as an accelerator?

Photo: HCHE
Centralized authorization procedures
In the EU, the European Medicines Agency (EMA, founded 1995) offers centralized authorization procedures to ensure the quality, safety and efficacy of pharmaceuticals. The EMA procedure can be either mandatory or voluntary. Successful applications allow market access to all countries covered by the EMA.

Photo: HCHE
Impact of EU membership
We examined whether EU membership has an impact on access to and availability of pharmaceuticals, using the accession countries of the EU's eastward expansion. In focus: 558 unique new active substances that were launched in Europe between 1998 and 2018.
*Excluding the following drugs: highly innovative and biotechnologically manufactured products, therapies for rare diseases (orphan drugs), active ingredients for the treatment of cancer, HIV/AIDS, diabetes, neurodegenerative or autoimmune diseases

Photo: HCHE
Recommendations for Further Acceleration
- Broadening the compulsory scope by including more substances that currently fall in the voluntary scope.
- In particular for those that are financially less attractive and less prioritized by manufacturers.
Where in Europe do patients wait the longest for new drugs?

Photo: HCHE
Delays in market entry in EU member states
There is a delay of between 0 and 25 months in the market launch across European member states.


